Methodology for characterizing network activations with neuro-navigated TMS and EEG
Recep Ozdemir, Emiliano Santarnecchi and Mouhsin M. Shafi
Berenson-Allen Center for Noninvasive Brain Stimulation, Beth Israel Deaconess Medical Center, Boston, MA (USA)
 This article walks users through the collection of neuro-navigated simultaneously obtained Transcranial Magnetic Stimulation (TMS) evoked EEG (TMS-EEG) data to investigate network-specific TMS-induced EEG activations, as in Ozdemir et al. 2020, focusing specifically on the methods of TMS-EEG data collection and analysis.
This article walks users through the collection of neuro-navigated simultaneously obtained Transcranial Magnetic Stimulation (TMS) evoked EEG (TMS-EEG) data to investigate network-specific TMS-induced EEG activations, as in Ozdemir et al. 2020, focusing specifically on the methods of TMS-EEG data collection and analysis.
To summarize that study, resting-state fMRI was used to define individualized targets for stimulation in nodes of the default mode network (DMN) and the dorsal attention network (DAN). Source-level EEG propagation patterns were evaluated, and found to be both network-specific and highly reproducible across sessions. Notably, the network specificity of TMS-evoked activity within the stimulated DMN versus DAN network, but not resting state dynamics, was correlated with individual differences in higher-order cognitive abilities, suggesting that TMS-EEG perturbational dynamics can characterize cognitively relevant network-level individual brain dynamics with high temporal resolution. The key methodology is summarized below, focusing on the collection and analysis of TMS-EEG data.
TMS Targets
A T1-weighted structural MRI scan with a spoiled gradient echo sequence was obtained for neuronavigation. Resting-state fMRI data were collected in three runs of 5 minutes each with eyes open. To identify individualized TMS targets, group-level resting-state functional network maps based on a seven-network parcellation (Yeo et al. 2011) were used. Confidence maps for each resting-state network were generated, with the confidence of each vertex indicating the likelihood of that vertex belonging to the assigned resting-state network in a sample of 1000 healthy participants. The seven resting state network parcellations and the group-level confidence maps were subsequently projected into each subject’s individual cortical surface using a spherical registration implemented in Freesurfer software, and resampled to the structural T1 SPGR sequences. The vertices with the highest confidence in the right angular gyrus were chosen for DMN stimulation, and the vertices with the highest confidence in the right superior parietal lobule were chosen for DAN stimulation. See Ozdemir et al. 2020 for further details.
TMS-EEG Data Collection Procedures
TMS is delivered using a figure-of-eight−shaped coil with dynamic fluid cooling (MagPro 75-mm Cool B-65; MagVenture A/S) attached to a MagPro X-100 stimulator (MagVenture A/S). Individual high-resolution T1w SPGR images are imported into the Brainsight TMS Frameless Navigation system (Rogue Research Inc.), and co-registered to digitized anatomical landmarks for online monitoring of coil positioning. Whole-scalp 64-channel EEG data were collected with a TMS-compatible amplifier system (actiCHamp system; Brain Products GmbH) and labeled in accordance with the extended 10–20 International system. The details of the TMS-EEG data collection are below.
TMS-EEG Visit Setup
Prior to the beginning of the TMS session, it is necessary to ensure that TMS is safe for the subject, ideally via a structured questionnaire. It is also beneficial to ensure that the subject did not engage in activities (e.g. experience significant sleep deprivation) or consume medications/substances (significant alcohol intake the prior night; extra caffeine; sodium channel blocking medications) that might alter cortical excitability.
In order to get a high-quality TMS-EEG recording, several steps are taken to ensure data quality. First, we need to properly set up the EEG system.
EEG Cap Fitting
 Prior to cap fitting, we clean the subject’s scalp and forehead with alcohol to remove any debris and oil. We then measure the head circumference (cm) to choose the appropriate EEG cap size. Once the cap is on the subject, measurements are taken from nasion to inion and from left and right preauricular points (cm) to determine the correct location of Cz. Cz should be in the midpoint of the distances nasion – inion and left to right preauricular. The ground, electrodes 1-32 and 33-64 are then plugged into their respective EEG amplifier ports, and then impedances are measured. All the lights on the cap’s electrodes that helpfully demonstrate the impedances should light up red, indicating an impedance of greater than 60 kOhm. For TMS-EEG experiments, to minimize the TMS pulse artifacts, the goal impedance is below 5 kOhm; if the impedance light range is set appropriately, this can be indicated by the green light.
Prior to cap fitting, we clean the subject’s scalp and forehead with alcohol to remove any debris and oil. We then measure the head circumference (cm) to choose the appropriate EEG cap size. Once the cap is on the subject, measurements are taken from nasion to inion and from left and right preauricular points (cm) to determine the correct location of Cz. Cz should be in the midpoint of the distances nasion – inion and left to right preauricular. The ground, electrodes 1-32 and 33-64 are then plugged into their respective EEG amplifier ports, and then impedances are measured. All the lights on the cap’s electrodes that helpfully demonstrate the impedances should light up red, indicating an impedance of greater than 60 kOhm. For TMS-EEG experiments, to minimize the TMS pulse artifacts, the goal impedance is below 5 kOhm; if the impedance light range is set appropriately, this can be indicated by the green light.
For TMS-EEG experiments, the selected reference and ground electrodes should be as far from the stimulation coil as possible to minimize the possibility of TMS-induced electrode artifacts contaminating the entire recording, and should ideally be placed above “inactive” structures with minimal cortical activity (although this may not always be possible or desirable). We start by filling the ground and reference electrodes with a small amount of conductive gel using a syringe applicator, and then rotate the applicator with some pressure to mildly abrade the skin and spread the gel to ensure that the volume between the electrodes and the skin is completely filled. Once the impedances are below 5 kOhm, the light on the electrode should turn green. During this process, please check in with the subject to ensure that they remain comfortable. Subsequently, all other electrodes can be prepared. Once all the electrodes are filled with gel, apply plastic wrap then gauze wrapping to secure the cap and keep gel moist.
Tips:
- If the reference and ground do not have low impedances, then the entire recording will be of low quality; it is essential to adequately prep these electrodes.
- Avoid putting an excess amount of gel in the electrodes to prevent leakage (bridging) between electrodes.
EMG Placement
In this experiment, TMS was administered at 120% resting motor threshold. Identification of resting motor threshold requires use of EMG recordings from the left first dorsal interosseous (FDI) and abductor pollicis brevis (APB) muscles (contralateral to the stimulated hemisphere). EMG activity is recorded using EMG boxes that are inserted into the AUX ports of the actiCHamp amplifier. Similar to the preparation of the EEG cap, it is necessary to wipe down the specific areas of interest with alcohol to remove any debris and oil on the skin. Then, prepare the skin with an abrasive agent (e.g. NuPrep) to exfoliate the skin and improve contact. ABRALYT-HiCl gel is then placed on to the EMG electrode and the electrode is secured with tape on to the belly and tendon of the target muscles (see image below for details).
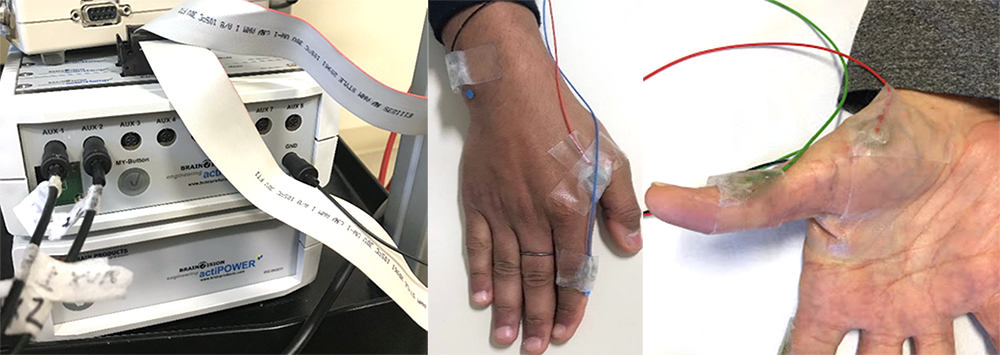
Neuronavigation Setup
Place the neuronavigation trackers on the participant’s head in a position that will be easily visible by the Polaris camera and register them on Brainsight using the pointer. Record the location of standard anatomic fiducial landmarks (e.g. nasion, left and right tragus) to co-register the subject’s head with the MRI, to enable neuronavigated stimulation of the target sites. Digitize and record the position of each EEG electrode by applying the pointer to the gap area of each electrode, and then holding the pointer steady when the sample is being taken.
Subject Positioning
Throughout the session, subjects are comfortably seated in an adjustable chair. For determining motor threshold, the subject is given a pillow to place in their lap, and instructed to keep both hands totally relaxed and rested on the pillow.
TMS-EEG Experimental Procedures
At the beginning of the session, it is often helpful to perform a recording of various artifacts. During this, the subject is asked to do a few movements (eye blinks, moving eyes left & right, moving eyes up & down, clenching jaw, chewing motion, wrinkling forehead) while EEG is recorded. This is to make sure the EEG recording system is functioning properly; to identify specific EEG correlates of various movements, which can be helpful in subsequent preprocessing; and to show the subject how sensitive the EEG recording is to various movements, which can help encourage them to remain in an alert, relaxed position throughout the session.
Minimizing Peripheral-evoked Potentials
TMS generates a clicking noise that can produce an auditory-evoked potential; TMS also generates a tapping sensation, and can produce muscle contractions underneath the coil, which can generate somatosensory-evoked potentials (Nikouline, Ruohonen, and Ilmoniemi 1999; Conde et al. 2019; Biabani et al. 2019; Rocchi et al. 2020). These peripheral-evoked potentials can overlap or even dominate the transcranial-evoked potentials, and thus appropriate steps need to be taken to try to minimize these elements. Research into this is ongoing, but current recommendations include continuously playing a masking noise, ideally white noise mixed with specific time-varying frequencies of the TMS click (Massimini et al. 2005; Rocchi et al. 2020), and with a thin layer of foam placed underneath the coil to minimize bone conduction and somatosensory sensations (ter Braack, de Vos, and van Putten 2015; Rocchi et al. 2020). Signal processing methods such as independent component analysis (ICA) or signal-space projection with source informed reconstruction can be used after the experimental session to minimize residual peripheral-evoked potentials (Biabani et al. 2019) although the efficacy of these post-hoc methods is debated.
Resting Motor Threshold Assessment
At the beginning of each session, the resting motor threshold is determined using the relative frequency method (Rossini et al. 2015). Briefly, the motor hotspot for eliciting MEPs in the FDI muscle is determined by delivering single TMS pulses over the hand region of left motor cortex with the TMS coil oriented approximately 45% posterolaterally in relation to the parasagittal plane (inducing posterior-to-anterior current in the underlying cortex). The TMS coil is moved in small incremental steps after two to three stimulations in each spot, and the hotspot is defined as the region where single-pulse TMS elicits larger and more consistent MEPs in the FDI muscle, as compared to the APB muscle. The FDI hotspot is digitized on each participant’s MRI image. Resting motor threshold (RMT) is determined on the FDI hotspot as the minimum stimulation intensity eliciting MEPs ≥50 μV on at least 5/10 trials.
Stimulation of DAN/DMN Nodes
A total of 120 single TMS pulses are delivered to each stimulation target (DMN node target in the angular gyrus and DAN node target in the superior parietal lobule). In the paper of Ozdemir et al. 2020, stimulation was applied at an intensity of 120% RMT with randomly jittered (3,000 to 5,000 ms) interstimulus intervals over two repeated blocks of 60 trials each. Note that other studies use shorter interstimulus intervals (e.g. 2000 to 2300 ms (Massimini et al. 2005); 1800 to 2200 ms (Kerwin et al. 2018)).
Tips:
- Make sure to monitor participants during the TMS blocks to ensure that they remain awake and relaxed; prompt them to keep their eyes open and remind them to relax the relevant muscles in case of either visible signs of drowsiness or tension, or if corresponding artifacts (slow roving eye movements, slowing, muscle artifacts) are seen in the EEG recording.
- The optimum method and intensity for stimulating non-motor regions has not been definitively established. Some authors suggest that differences in scalp-cortex distance across brain regions should be adjusted for (Stokes et al. 2007), or, in neuronavigation systems capable of performing online estimations of the induced electric field, the intensity of the stimulation can be set at a specific amplitude of the calculated induced electric field (in V/m) on the cortical surface (Rosanova et al. 2009).
Sham Stimulation
A sham stimulation session is recommended to provide supportive evidence that the noise masking is effective, and to help control/identify and remove residual peripheral-evoked potentials during postprocessing. Specific coils are available (e.g. Magventure Cool-B65 A/P coil) in which effective stimulation is delivered on one side, while the magnetic field is markedly attenuated (e.g. 5% of active side) on the placebo side. Administration from the sham side with a spacer can help diminish the induced electric field in the cortex to essentially zero. An adequate sham stimulation should include the auditory “click” sound produced by real TMS, and should be coupled with electrical stimulation under the coil in order to induce somatosensory sensations similar to real TMS (Conde 2019; Rocchi 2020); however, it is worth noting that the sensation induced by electrical stimulation is not identical to that produced by TMS (Conde 2019).
TMS-EEG data processing
Preprocessing
TMS-EEG data requires preprocessing to obtain clean TMS-evoked EEG potentials. In the study of Ozdemir et al. 2020, TMS-EEG preprocessing was conducted using the free MATLAB® (Mathworks, Inc.) software EEGLAB (Delorme and Makeig 2004) with the TMS-EEG Signal Analyzer (TESA) extension (Rogasch et al. 2017). The two separate blocks of 60 trials from each stimulation site were merged together into a single block of 120 trials. The merged trials were then segmented into 3000 ms epochs starting 1000 ms before and ending 2000 ms after the TMS pulse. Following baseline correction of the resulting epochs, noisy channels were identified and deleted by visual inspection. The epochs were zero-padded from 2 ms before to 14 ms after the TMS pulse to remove the early TMS pulse artifact. The resulting zero-padded epochs were tagged based on voltage, kurtosis and probability measures, and the resulting data visually inspected to identify and remove noisy epochs. Next, an initial round of fast ICA was completed to exclusively identify and remove components with high-amplitude early TMS-evoked artifacts. Subsequently, EEG data were interpolated during the previously zero-padded time window from -2 to 14 ms, bandpass filtered, and re-referenced to global average. Subsequently, a second round of ICA was performed to remove artifacts related to eye blinks, eye movements, muscle, cardiac (EKG), and single electrode noise, as well as residual TMS artifacts and auditory evoked potentials. Finally, the resulting data was low-pass filtered, and missing channels were interpolated using a spherical spline interpolation to obtain the final TMS-evoked EEG potentials.
Source Analysis
The cortical source activations corresponding to the scalp TMS-evoked EEG potentials were calculated to determine the brain network-specific activations with TMS stimulation. Source analysis was performed using Brainstorm (Tadel et al. 2011). Digitized EEG channel locations and the anatomical landmarks for each subject were registered into individual MRI scans in Brainstorm. The average TMS-evoked potential time series was obtained for each subject. Forward modeling to determine the electric field from each vertex was performed using the symmetric boundary element method (Gramfort et al. 2010). The noise covariance was estimated using the 500 ms pre-TMS as the baseline. Inverse modeling was performed using the minimum noise estimation (Hämäläinen and Ilmoniemi 1994) method with dynamical statistical parametric mapping and with source dipoles constrained to the cortical surface. The resulting output was the current density time series at each cortical vertex. To obtain network activations, the average current density time series was obtained across two regions-of-interest, defined as the projections of the default mode network and dorsal attention network onto each individual surface space. The current densities were z-scored normalized to the baseline, rectified, and summed to calculate the total amount of source reconstructed cortical activity in each resting-state network map.
Conclusion
Using the above approach, it is possible to characterize the TMS-evoked activations within distinct resting-state fMRI-defined cortical networks. By applying this approach with stimulation of individualized nodes of the default mode and dorsal attention networks, Ozdemir et al. 2020 found that TMS-evoked activity is both network-specific and highly reproducible across sessions, and that the degree of this network specificity was correlated with individual higher-order cognitive abilities. More broadly, this approach could be utilized to identify TMS-evoked activity within any cortical region of interest, and thus study the effective network connectivity of different cortical targets.
References
Biabani, Mana, Alex Fornito, Tuomas P. Mutanen, James Morrow, and Nigel C. Rogasch. 2019.
“Characterizing and Minimizing the Contribution of Sensory Inputs to TMS-Evoked Potentials.”
Brain Stimulation 12 (6): 1537–52. https://doi.org/10.1016/j.brs.2019.07.009.Braack, Esther M. ter, Cecile C. de Vos, and Michel J. A. M. van Putten. 2015.
“Masking the Auditory Evoked Potential in TMS-EEG: A Comparison of Various Methods.”
Brain Topography 28 (3): 520–28. https://doi.org/10.1007/s10548-013-0312-z.Conde, Virginia, Leo Tomasevic, Irina Akopian, Konrad Stanek, Guilherme B. Saturnino, Axel Thielscher, Til Ole Bergmann, and Hartwig Roman Siebner. 2019.
“The Non-Transcranial TMS-Evoked Potential Is an Inherent Source of Ambiguity in TMS-EEG Studies.”
NeuroImage 185: 300–312. https://doi.org/10.1016/j.neuroimage.2018.10.052.Delorme, Arnaud, and Scott Makeig. 2004.
“EEGLAB: An Open Source Toolbox for Analysis of Single-Trial EEG Dynamics Including Independent Component Analysis.”
Journal of Neuroscience Methods 134 (1): 9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009.Gramfort, Alexandre, Théodore Papadopoulo, Emmanuel Olivi, and Maureen Clerc. 2010.
“OpenMEEG: Opensource Software for Quasistatic Bioelectromagnetics.”
Biomedical Engineering Online 9 (September): 45. https://doi.org/10.1186/1475-925X-9-45.Hämäläinen, M. S., and R. J. Ilmoniemi. 1994.
“Interpreting Magnetic Fields of the Brain: Minimum Norm Estimates.”
Medical & Biological Engineering & Computing 32 (1): 35–42. https://doi.org/10.1007/BF02512476.Kerwin, Lewis J., Corey J. Keller, Wei Wu, Manjari Narayan, and Amit Etkin. 2018.
“Test-Retest Reliability of Transcranial Magnetic Stimulation EEG Evoked Potentials.”
Brain Stimulation 11 (3): 536–44. https://doi.org/10.1016/j.brs.2017.12.010.Massimini, Marcello, Fabio Ferrarelli, Reto Huber, Steve K. Esser, Harpreet Singh, and Giulio Tononi. 2005.
“Breakdown of Cortical Effective Connectivity during Sleep.”
Science (New York, N.Y.) 309 (5744): 2228–32. https://doi.org/10.1126/science.1117256.Nikouline, V., J. Ruohonen, and R. J. Ilmoniemi. 1999.
“The Role of the Coil Click in TMS Assessed with Simultaneous EEG.”
Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology 110 (8): 1325–28. https://doi.org/10.1016/s1388-2457(99)00070-x.Ozdemir, Recep A., Ehsan Tadayon, Pierre Boucher, Davide Momi, Kelly A. Karakhanyan, Michael D. Fox, Mark A. Halko, Alvaro Pascual-Leone, Mouhsin M. Shafi, and Emiliano Santarnecchi. 2020.
“Individualized Perturbation of the Human Connectome Reveals Reproducible Biomarkers of Network Dynamics Relevant to Cognition.”
Proceedings of the National Academy of Sciences of the United States of America 117 (14): 8115–25. https://doi.org/10.1073/pnas.1911240117.Rocchi, Lorenzo, Alessandro Di Santo, Katlyn Brown, Jaime Ibáñez, Elias Casula, Vishal Rawji, Vincenzo Di Lazzaro, Giacomo Koch, and John Rothwell. 2020.
“Disentangling EEG Responses to TMS Due to Cortical and Peripheral Activations.”
Brain Stimulation 14 (1): 4–18. https://doi.org/10.1016/j.brs.2020.10.011.Rogasch, Nigel C., Caley Sullivan, Richard H. Thomson, Nathan S. Rose, Neil W. Bailey, Paul B. Fitzgerald, Faranak Farzan, and Julio C. Hernandez-Pavon. 2017.
“Analysing Concurrent Transcranial Magnetic Stimulation and Electroencephalographic Data: A Review and Introduction to the Open-Source TESA Software.”
NeuroImage 147: 934–51. https://doi.org/10.1016/j.neuroimage.2016.10.031.Rosanova, Mario, Adenauer Casali, Valentina Bellina, Federico Resta, Maurizio Mariotti, and Marcello Massimini. 2009.
“Natural Frequencies of Human Corticothalamic Circuits.”
The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 29 (24): 7679–85. https://doi.org/10.1523/JNEUROSCI.0445-09.2009.Rossini, P. M., D. Burke, R. Chen, L. G. Cohen, Z. Daskalakis, R. Di Iorio, V. Di Lazzaro, et al. 2015.
“Non-Invasive Electrical and Magnetic Stimulation of the Brain, Spinal Cord, Roots and Peripheral Nerves: Basic Principles and Procedures for Routine Clinical and Research Application. An Updated Report from an I.F.C.N. Committee.”
Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology 126 (6): 1071–1107. https://doi.org/10.1016/j.clinph.2015.02.001.Stokes, Mark G., Christopher D. Chambers, Ian C. Gould, Therese English, Elizabeth McNaught, Odette McDonald, and Jason B. Mattingley. 2007.
“Distance-Adjusted Motor Threshold for Transcranial Magnetic Stimulation.”
Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology 118 (7): 1617–25. https://doi.org/10.1016/j.clinph.2007.04.004.Tadel, François, Sylvain Baillet, John C. Mosher, Dimitrios Pantazis, and Richard M. Leahy. 2011.
“Brainstorm: A User-Friendly Application for MEG/EEG Analysis.”
Computational Intelligence and Neuroscience 2011: 879716. https://doi.org/10.1155/2011/879716.Yeo, B. T. Thomas, Fenna M. Krienen, Jorge Sepulcre, Mert R. Sabuncu, Danial Lashkari, Marisa Hollinshead, Joshua L. Roffman, et al. 2011.
“The Organization of the Human Cerebral Cortex Estimated by Intrinsic Functional Connectivity.”
Journal of Neurophysiology 106 (3): 1125–65. https://doi.org/10.1152/jn.00338.2011

