Simultaneous EEG-fMRI provides a new insight into the origin of spontaneous neuronal activity in preterm humans
by Tomoki Arichi1,2 and Lorenzo Fabrizi3
1Centre for the Developing Brain, Division of Imaging Sciences and Biomedical Engineering, King’s College London, St Thomas’ Hospital, London, SE1 7EH, UK
2Department of Bioengineering, Imperial College London, South Kensington Campus, London, SW7 2AZ, UK
3Department of Neuroscience, Physiology & Pharmacology, University College London, Gower Street, London, WC1 E6BT, UK
Acknowledgement
This user research article summarizes our publication “Arichi T, Whitehead K, Barone G, Pressler R, Padormo F, Edwards AD, Fabrizi L (2017). Localization of spontaneous bursting neuronal activity in the preterm human brain with simultaneous EEG-fMRI. eLife 6:e27814”.
Introduction
During the third trimester of human gestation (28 to 40 weeks post-menstrual age (PMA)), the brain undergoes a sequence of functional and structural maturational changes as the cortex and its underlying framework of connectivity are established. The importance of this period is emphasised by the greater risk of adverse neurodevelopmental outcome in infants delivered preterm (prior to 37 weeks PMA) [1, 2].
In animal models, spontaneous bursts of synchronized activity (known as spindle bursts) play an instructive role in the developmental processes that set early cortical circuits [3-5]. In keeping with this, experimental disruption of the normal occurrence and propagation of early spontaneous patterned activity leads to permanent loss of healthy cortical organization [6, 7]. Similarly, neural activity recorded in preterm human infants with electroencephalography (EEG) is characterized by intermittent high amplitude bursts which can occur both spontaneously and following external stimulation [4, 8, 9]. The most common of these events is the delta brush, a transient pattern characterised by a slow delta wave (1-4Hz) with a superimposed fast frequency spindle (8-25Hz) [8]. This pattern appears to represent a key marker of healthy neuronal development as their persistence at term equivalent age (or an earlier absence) correlates with underlying brain injury and/or adverse neurological outcome later in childhood [10]. However, despite the common occurrence, developmental importance and clinical significance of delta brushes, the brain structures which generate this hallmark neuronal pattern within the developing human brain remain unknown.
While the time of occurrence and the scalp distribution of a delta brush event can be readily identified with EEG, the spatial localization of its source cannot be inferred with certainty just from the electrical potentials recorded at the scalp surface. To overcome this intrinsic limitation of EEG recording, we pioneered the use of simultaneous EEG-fMRI in human neonates. Recent research has explored the direct correlation between neural and hemodynamic function in adult humans with this method, demonstrating the potential insights it can provide in both health and pathology. For example, EEG-fMRI has been employed to identify the source of epileptogenic EEG activity in presurgical planning [11], to investigate the association between spontaneous EEG oscillations and fMRI fluctuations at rest [12] or in cognitive neuroscience to localise the source of task-related event related potentials [13, 14]. However, the coupling between resting neuronal and hemodynamic activity has never been explored in the developing infant brain. Here, we provide the first evidence that spontaneous patterns of delta brush activity in the preterm period are associated with significant hemodynamic activity clearly localized to distinct regions within the developing cortex.
Methods
Participants
Thirteen preterm infants (five females; studied between 32-36 weeks post-menstrual age, PMA) 5-55 days old (23 ± 17, mean ± SD) from the Neonatal Unit at St Thomas’ Hospital, London, participated in this study. Informed written parental consent was obtained prior to each study. All of the research methods conformed to the standards set by the Declaration of Helsinki and was approved by the National Research Ethics Committee.
EEG-fMRI acquisition
MR images were collected during sleep on a 3-Tesla Philips Achieva scanner (Best, Netherlands). Concurrent EEG recording were conducted with a custom-made BrainCap MR sized for the head of preterm infant containing 26-32 scalp electrodes (EASYCAP GmbH, DE) connected to an MR-compatible EEG system (BrainAmp MR, Brain Products GmbH, DE).
EEG-fMRI pre-processing and analysis
Gradient artefacts caused by the MR image acquisition in the EEG were corrected using Analyzer 2 software (Brain Products, DE). The typical EEG cardioballistic artefacts were not present in our neonatal recordings. fMRI standard pre-processing was performed using FSL (FMRIB’s software library, www.fmrib.ox.ac.uk/fsl) [15]. Residual motion and physiological noise were removed with Probabilistic Independent Component Analysis [PICA, v3.0]) [16].
Three independent trained observers reviewed the EEG recordings and marked the occurrence of delta brush events on the same software. Events were then labelled based on their field distribution and used as separate Explanatory Variables (EVs) in the general linear model (GLM) of the fMRI analysis to generate spatial maps of activated voxels at an individual subject level. Only EVs containing more than 3 events during the acquisition period were used for analysis. Individual subject activation maps were then co-aligned to an age-specific neonatal atlas for group analysis.
Results
Simultaneous EEG-fMRI was successfully acquired in 10 of the 13 infants over a median of 7.5 minutes (range: 3.5-10.5 minutes).
A median of 4.4 delta brushes per minute (range: 1.9 – 6.7) with a total of 23 distinct topographical distributions were found across all subjects. However, only 10 topographies occurred more than three times in a given subject and were included in the fMRI analysis (Figure 1). These covered the parietal, occipital and temporal, but not frontal areas of the scalp.
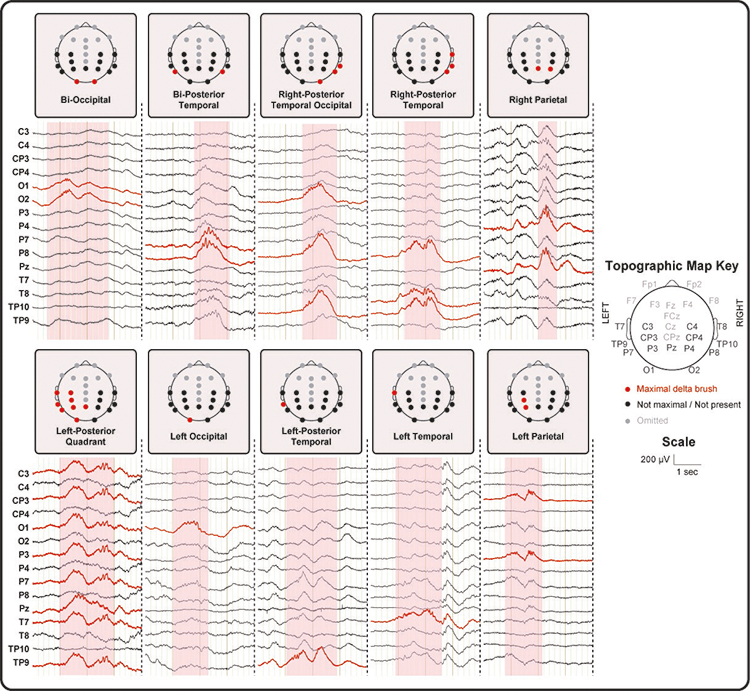
Figure 1: Examples of delta brushes with different topographies recorded in the MR scanner and after gradient artefact removal.
We then found that different delta brush topographies were associated with different BOLD activity maps at subject level. When significant areas of BOLD activity were present, this always included at least one cluster ipsilateral to the side of the recorded delta brush activity. As most of the delta brushes in our study consistently occurred over the left (n = 78 from 9/10 subjects) and right (n = 86 from 10/10 subjects) posterior-temporal regions, the corresponding single subject data were taken forward to group analysis. Lateralized posterior-temporal delta brush activity was associated on both a single subject and group level with significant clusters of positive BOLD activity in the insular cortex ipsilateral to the delta brush activity (Figure 2). Right-sided delta brush activity was also associated with significant clusters of hemodynamic activity in the right superior temporal cortex and extending to the right temporal pole (Figure 2).
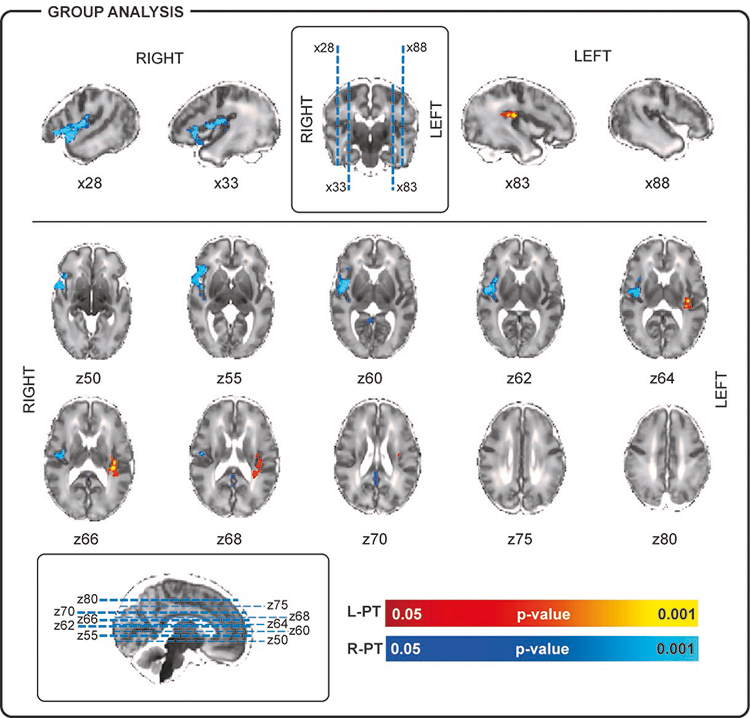
Figure 2: BOLD activity associated with the occurrence of left posterior-temporal (L-PT) and right posterior-temporal (R-PT) delta brushes.
Discussion
The human preterm EEG is characterised by spontaneous neuronal bursts, which in animals are known to be crucial for cortical development. Here we have shown that these are organized into a set of spatiotemporal patterns, the most common of which has a unilateral posterior-temporal scalp distribution in the period equivalent to the last trimester of gestation. These bursts are associated with ipsilateral clusters of hemodynamic activation in the insular cortex only and insular and temporal cortices when occurring over the left and right hemispheres respectively.
As hypothesised, the simultaneous acquisition of EEG-fMRI data proved to be highly complementary, permitting the source localization of the delta brush activity, not previously achieved. By taking advantage of natural sleep, we were able to successfully record data for analysis in 10 of the 13 subjects studied. This approach allowed us to identify specific developmental patterns of spontaneous neural activity with EEG and subsequently link this activity directly to the whole-brain spatial information offered by fMRI, combining the strengths of these two techniques and overcoming their individual limitations.
The incidence and topographical distribution of the delta brush changes across the preterm period. Over this period, delta brushes can also be elicited by external sensory stimuli [10] and their topography is related to the stimulus modality: visual, auditory and tactile stimulation evoke occipital, mid-temporal and pericentral delta brushes [17-19]. These observations suggest that delta brushes with different topographies (whether spontaneous or evoked) are related to distinct brain functions which develop in different time frames. Our discovery here, that spontaneous posterior-temporal delta brushes arise in the insula and temporal pole, has therefore important implications for understanding the events underlying brain development in the preterm infant.
The mature insula is a structural and functional hub with a dense pattern of connectivity to almost all other regions of the brain, enabling it to play a versatile role in a wide range of functions including sensory and pain perception, emotion, and cognition [20]. As in humans, the insula plays an important multisensory role also in adult rodents [21]. This function develops in the third postnatal week (equivalent to the late preterm period in humans) with the maturation of inhibitory circuits and an optimal excitation/inhibition balance [22], however nothing is known about its spontaneous activity as most of these kind of recordings have been from the primary sensory cortices. Our results suggest that it could be important to record from this area as it may represent a major source of early neuronal bursting activity in rodents as well as in humans.
Our findings are of further significance in light of the increasing number of studies using fMRI to characterize developmental changes in resting state functional connectivity and task-induced responses in early infancy [23]. Despite marked maturational changes in early life in the neurovascular coupling cascade [24, 25], we demonstrate for the first time in this population, a clear association between a direct measure of neural activity (EEG) and functional hemodynamic activity as measured by fMRI. This provides the potential to understand hemodynamic fluctuations in terms of the underlying neuronal rhythms and, in particular, to link connectivity measures across the two modalities [26]. Although the majority of resting state networks at term equivalent age are spatially similar to those observed in adults [27, 28] their role and underlying neuronal processes may differ.
This study provides the first evidence that spontaneous bursting neuronal activity, and specifically the delta brush, is largely generated by the insula in the late human preterm period. The insula is a phylogenetically ancient part of the brain that undergoes dramatic functional and structural maturation at this time, preceding that of the overlying neocortex. Since the equivalent of the delta brush in rodents has an instructive function in the normal neuronal maturation of the cortex, we propose that the insula plays a key developmental role as generator of such activity in early human life.
References
[1] 1. Allen, M.C. (2008).
Neurodevelopmental outcomes of preterm infants.
Curr. Opin. Neurol. 21, 123-128.[2] Larroque, B., Ancel, P.Y., Marret, S., Marchand, L., Andre, M., Arnaud, C., Pierrat, V., Roze, J.C., Messer, J., Thiriez, G., et al. (2008).
Neurodevelopmental disabilities and special care of 5-year-old children born before 33 weeks of gestation (the EPIPAGE study): a longitudinal cohort study. Lancet 371, 813-820.[3] Hanganu-Opatz, I.L. (2010).
Between molecules and experience: role of early patterns of coordinated activity for the development of cortical maps and sensory abilities.
Brain Res Rev 64, 160-176.[4] Khazipov, R., and Luhmann, H.J. (2006).
Early patterns of electrical activity in the developing cerebral cortex of humans and rodents.
Trends Neurosci. 29, 414-418.[5] Rakic, P., and Komuro, H. (1995).
The role of receptor/channel activity in neuronal cell migration.
J. Neurobiol. 26, 299-315.[6] Xu, H.P., Furman, M., Mineur, Y.S., Chen, H., King, S.L., Zenisek, D., Zhou, Z.J., Butts, D.A., Tian, N., Picciotto, M.R., et al. (2011).
An Instructive Role for Patterned Spontaneous Retinal Activity in Mouse Visual Map Development.
Neuron 70, 1115-1127.[7] Tolner, E.A., Sheikh, A., Yukin, A.Y., Kaila, K., and Kanold, P.O. (2012).
Subplate neurons promote spindle bursts and thalamocortical patterning in the neonatal rat somatosensory cortex.
J. Neurosci. 32, 692-702.[8] Andre, M., Lamblin, M.D., d’Allest, A.M., Curzi-Dascalova, L., Moussalli-Salefranque, F., T, S.N.T., Vecchierini-Blineau, M.F., Wallois, F., Walls-Esquivel, E., and Plouin, P. (2010).
Electroencephalography in premature and full-term infants. Developmental features and glossary.
Neurophysiol. Clin. 40, 59-124.[9] Tolonen, M., Palva, J.M., Andersson, S., and Vanhatalo, S. (2007).
Development of the spontaneous activity transients and ongoing cortical activity in human preterm babies.
Neuroscience 145, 997-1006.[10] Whitehead, K., Pressler, R., and Fabrizi, L. (2017).
Characteristics and clinical significance of delta brushes in the EEG of premature infants.
Clinical Neurophysiology Practice 2, 12-18.[11] Gotman, J., and Pittau, F. (2011).
Combining EEG and fMRI in the study of epileptic discharges.
Epilepsia 52 Suppl 4, 38-42.[12] Laufs, H. (2008).
Endogenous brain oscillations and related networks detected by surface EEG-combined fMRI.
Hum. Brain Mapp. 29, 762-769.[13] Huster, R.J., Debener, S., Eichele, T., and Herrmann, C.S. (2012).
Methods for simultaneous EEG-fMRI: an introductory review.
J. Neurosci. 32, 6053-6060.[14] Ritter, P., and Villringer, A. (2006).
Simultaneous EEG-fMRI.
Neurosci. Biobehav. Rev. 30, 823-838.[15] Smith, S.M., Jenkinson, M., Woolrich, M.W., Beckmann, C.F., Behrens, T.E., Johansen-Berg, H., Bannister, P.R., De Luca, M., Drobnjak, I., Flitney, D.E., et al. (2004).
Advances in functional and structural MR image analysis and implementation as FSL.
Neuroimage 23 Suppl 1, S208-219.[16] Beckmann, C.F., and Smith, S.M. (2004).
Probabilistic independent component analysis for functional magnetic resonance imaging.
IEEE Trans. Med. Imaging 23, 137-152.[17] Chipaux, M., Colonnese, M.T., Mauguen, A., Fellous, L., Mokhtari, M., Lezcano, O., Milh, M., Dulac, O., Chiron, C., Khazipov, R., et al. (2013).
Auditory Stimuli Mimicking Ambient Sounds Drive Temporal “Delta-Brushes” in Premature Infants.
PloS one 8.[18] Colonnese, M.T., Kaminska, A., Minlebaev, M., Milh, M., Bloem, B., Lescure, S., Moriette, G., Chiron, C., Ben-Ari, Y., and Khazipov, R. (2010).
A conserved switch in sensory processing prepares developing neocortex for vision.
Neuron 67, 480-498.[19] Milh, M., Kaminska, A., Huon, C., Lapillonne, A., Ben-Ari, Y., and Khazipov, R. (2007).
Rapid cortical oscillations and early motor activity in premature human neonate.
Cereb. Cortex 17, 1582-1594.[20] Nieuwenhuys, R. (2012).
The insular cortex: a review.
Prog. Brain Res. 195, 123-163.[21] Rodgers, K.M., Benison, A.M., Klein, A., and Barth, D.S. (2008).
Auditory, somatosensory, and multisensory insular cortex in the rat.
Cereb. Cortex 18, 2941-2951.[22] Gogolla, N., Takesian, A.E., Feng, G., Fagiolini, M., and Hensch, T.K. (2014).
Sensory integration in mouse insular cortex reflects GABA circuit maturation.
Neuron 83, 894-905.[23] Graham, A.M., Pfeifer, J.H., Fisher, P.A., Lin, W., Gao, W., and Fair, D.A. (2015).
The potential of infant fMRI research and the study of early life stress as a promising exemplar.
Developmental cognitive neuroscience 12, 12-39.[24] Arichi, T., Fagiolo, G., Varela, M., Melendez-Calderon, A., Allievi, A., Merchant, N., Tusor, N., Counsell, S.J., Burdet, E., Beckmann, C.F., et al. (2012).
Development of BOLD signal hemodynamic responses in the human brain.
Neuroimage 63, 663-673.[25] Harris, J.J., Reynell, C., and Attwell, D. (2011).
The physiology of developmental changes in BOLD functional imaging signals.
Developmental cognitive neuroscience 1, 199-216.[26] Vanhatalo, S., and Fransson, P. (2016).
Advanced EEG and MRI Measurements to Study the Functional Development of the Newborn Brain. In Prenatal and Postnatal Determinants of Development, D.W. Walker, ed. (New York: Springer New York), pp. 53-68.[27] Doria, V., Beckmann, C.F., Arichi, T., Merchant, N., Groppo, M., Turkheimer, F.E., Counsell, S.J., Murgasova, M., Aljabar, P., Nunes, R.G., et al. (2010).
Emergence of resting state networks in the preterm human brain.
Proc Natl Acad Sci U S A 107, 20015-20020.[28] Smyser, C.D., Inder, T.E., Shimony, J.S., Hill, J.E., Degnan, A.J., Snyder, A.Z., and Neil, J.J. (2010).
Longitudinal analysis of neural network development in preterm infants.
Cereb. Cortex 20, 2852-2862.

